Please note, the terms and conditions relating to service, product sales, promotions, and other related activities can be found at, and those terms and conditions control any purchases of products or services from the Company. These Terms and Conditions apply only to the use of the Web Site. Not all of the products or services described in this Web Site are available in all jurisdictions or to all potential customers, and nothing herein is intended as an offer or solicitation in any jurisdiction or to any potential customer where such offer or sale is not qualified. Although the information is believed to correct at the time of publishing, you should make your own determination as to its suitability for your use. The information contained on this Web Site is provided for informational purposes only. If you disagree with the Terms of Use (as amended from time to time) or are dissatisfied with the Web Site, your sole and exclusive remedy is to discontinue using the Web Site. Your continued use of the Web Site means that you accept and agree to the revised Terms of Use. The Company may revise and update these Terms of Use at any time without notice by posting the amended terms to the Web Site. If you do not wish to agree to these Terms of Use, do not access or use any part of the Web Site. By accessing or using the Web Site, you agree that you have read, understand and agree to be bound by these Terms of Use, as amended from time to time, as well as the Company Privacy Policy, which is hereby incorporated into these Terms of Use. Please read these Terms of Use carefully before accessing or using any part of the Web Site. All users of the Web Site are subject to the following website terms and conditions of use (these “Terms of Use”). 
If you are accessing the Web Site from outside the United States, Canada, or Puerto Rico, please see the appropriate international website, available at for applicable terms and conditions. These terms and conditions of use are applicable to the United States, Canada and Puerto Rico websites (“collectively the Web Site”) operated by VWR (the “Company”). HALO® Chromatography Columns and Consumables+. Sartorius laboratory instruments, consumables and services PerkinElmer - Innovating for a Healthier World Lab Thermometers & Temperature Measurement EquipmentĪgilent Chemistries and Supplies Portfolio Quality Products from Sheldon Manufacturing Life Science Research Solutions, Products, and Resources VWR will support you from the latest life science products to the guaranteed purity of organic building blocks. A mechanistic insight into the higher activity of complex 1 towards the OER compared to that of complex 2 is also provided using density functional theory based calculations.A strong, vibrant research and development group is the lifeblood of all industries. The turnover frequency of this catalyst was calculated to be 5.23 s −1 with an excellent faradaic efficiency of 97%, indicating the selective oxygen evolution reaction (OER) occurring with the aid of this catalyst. Excellent current densities of 1 and 10 mA cm −2 were achieved with overpotentials of 354 and 452 mV respectively. The tetranuclear catalyst has excellent electrochemical stability and longevity, as established by chronoamperometry and >1000 cycle durability tests under highly alkaline conditions. 1 has been found to be an efficient catalyst for electrochemical water oxidation under highly basic conditions while the mononuclear analogue, 2, does not respond to electrochemical water oxidation. In 2, the Co( II) centre is surrounded by two monoanionic − ligands and a pair of methanol and acetonitrile solvents in a six-coordinate arrangement. Changing the Co( II) precursor from Co(OAc) 2♴H 2O to Co(NO 3) 2♶H 2O afforded a mononuclear complex (MeOH) 2 ( 2). The fourth coordination site on Co( II) is taken by an acetonitrile ligand. 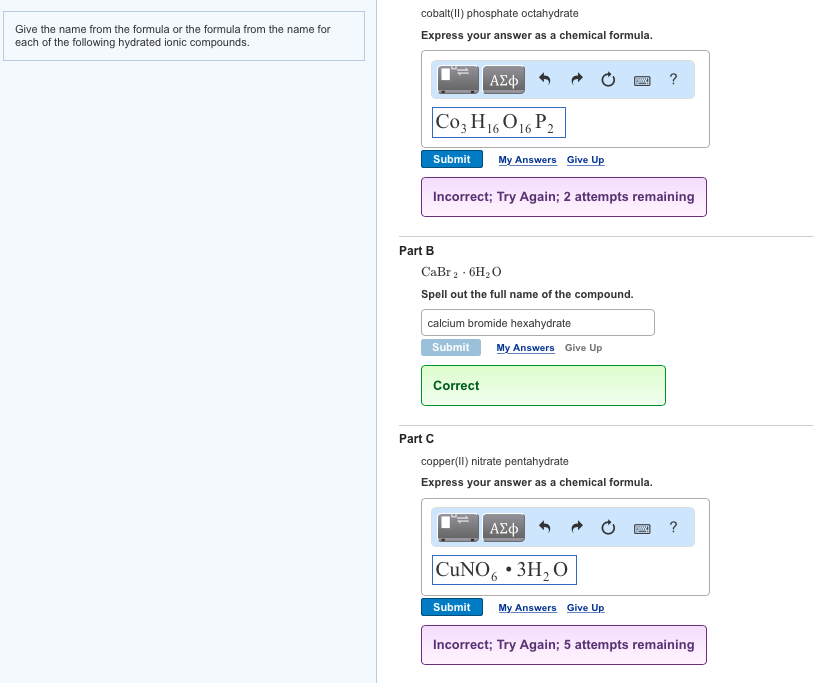
The four Co( II) centers are held together by four di-anionic 2− ligands. The molecular structure of 1 reveals that it is a tetranuclear assembly where the Co( II) centers are present in the alternate corners of a cube. The reaction of Co(OAc) 2♴H 2O with a sterically hindered phosphate ester, LH 2, afforded a tetranuclear complex, 4♵CH 3CN ( 1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
